Antibiotic resistance represents one of the most critical challenges in modern medicine and global public health. It refers to the ability of bacterial pathogens to survive and proliferate despite exposure to antimicrobial agents that were previously effective against them. This phenomenon is not new; however, its rapid acceleration in recent decades has transformed it into a persistent and complex global health threat.
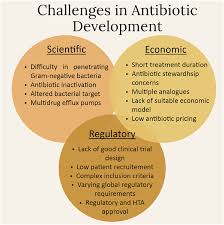
The rise of antibiotic resistance is driven by multiple interconnected factors, including inappropriate antibiotic use, lack of rapid diagnostic tools, insufficient infection control practices, and limited development of new antimicrobial drugs. Addressing this issue requires coordinated efforts across healthcare systems, policy frameworks, research institutions, and agricultural sectors.
Healthcare professionals, policymakers, and researchers are increasingly tasked with implementing effective strategies to slow down resistance development. These strategies include surveillance systems, antimicrobial stewardship programs, improved prescribing practices, and investment in novel therapeutic approaches.
Global Burden and Clinical Impact
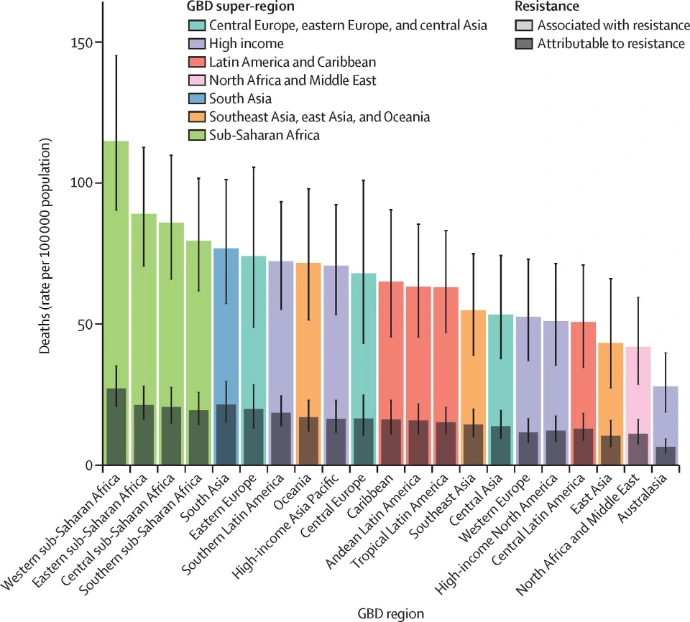
Antimicrobial resistance (AMR) in bacterial pathogens is associated with increased morbidity, mortality, and healthcare costs worldwide. Infections caused by multidrug-resistant (MDR) organisms are significantly more difficult to treat and often require prolonged hospitalization, intensive care, and the use of last-resort antibiotics.
Both Gram-positive and Gram-negative bacteria have developed resistance mechanisms that compromise the effectiveness of conventional treatments. As a result, many infections that were once easily manageable are now associated with poor clinical outcomes.
A major contributing factor to this problem is the widespread empirical use of broad-spectrum antibiotics, especially in settings where rapid diagnostic tools are unavailable. In many healthcare environments, clinicians initiate treatment without precise identification of the causative pathogen, leading to unnecessary or inappropriate antibiotic exposure. This practice accelerates the selection and spread of resistant bacterial strains.
Up-to-date epidemiological data on resistance patterns are essential for guiding treatment decisions and designing effective antimicrobial stewardship programs. Continuous monitoring allows healthcare systems to adapt therapeutic protocols and limit the spread of resistant organisms.
Mechanisms of Antibiotic Resistance
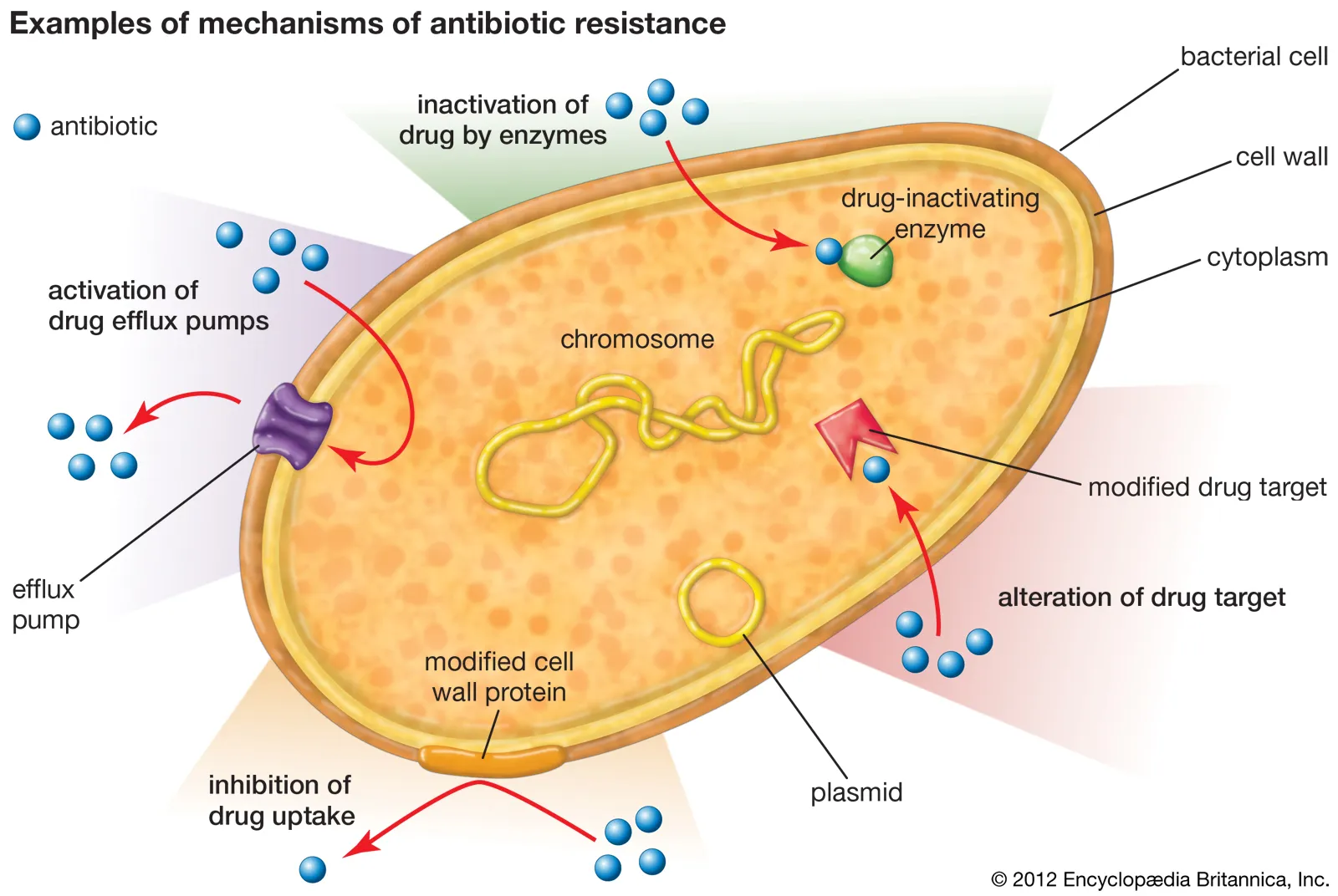
Bacteria have evolved multiple mechanisms to evade the effects of antimicrobial agents. These mechanisms can be intrinsic or acquired and often involve genetic adaptations that enhance bacterial survival.
Key resistance mechanisms include:
- Reduced drug uptake: Alterations in membrane permeability prevent antibiotics from entering the bacterial cell.
- Efflux pumps: Active transport systems expel antibiotics out of the cell before they can exert their effect.
- Enzymatic degradation: Production of enzymes such as beta-lactamases that inactivate antibiotics.
- Target modification: Structural changes in bacterial targets reduce antibiotic binding.
- Biofilm formation: Organized bacterial communities that provide physical and biochemical protection.
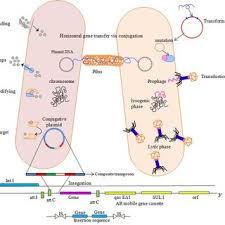
In addition to these mechanisms, bacteria can acquire resistance genes through horizontal gene transfer (HGT), which plays a central role in the dissemination of resistance across species and environments.
The Resistome and Gene Transfer
The concept of the resistome encompasses the entire collection of antibiotic resistance genes present in pathogenic, commensal, and environmental bacteria. This genetic reservoir serves as a source from which clinically relevant pathogens can acquire resistance traits.
Horizontal gene transfer occurs through several mechanisms:
- Conjugation: Transfer of genetic material via direct cell-to-cell contact.
- Transformation: Uptake of free DNA from the environment.
- Transduction: Transfer mediated by bacteriophages.
Biofilms and Their Role in Resistance
Biofilms are structured communities of microorganisms embedded in a self-produced extracellular matrix composed of polysaccharides, proteins, and DNA. This mode of growth significantly enhances bacterial survival under adverse conditions.
Biofilm-associated infections are particularly difficult to treat due to:
- Reduced antibiotic penetration
- Altered bacterial metabolism
- Increased genetic exchange
- Enhanced resistance to immune responses
Biofilms are commonly associated with chronic infections and medical devices such as catheters, implants, and prosthetics. They also play a critical role in diseases like chronic wounds and respiratory infections.
Standard antibiotic therapies are often ineffective against biofilms, requiring higher doses and prolonged treatment durations. However, these approaches frequently fail and contribute to further resistance development.
Emerging strategies to combat biofilms include:
- Disruption of the biofilm matrix
- Inhibition of quorum sensing (bacterial communication)
- Use of antimicrobial coatings on medical devices
- Development of biofilm-targeted therapeutics
Advances in Diagnostic Technologies
Rapid and accurate detection of bacterial pathogens and their resistance profiles is essential for effective treatment. Traditional diagnostic methods are often time-consuming and may delay appropriate therapy.
Recent technological advancements include:
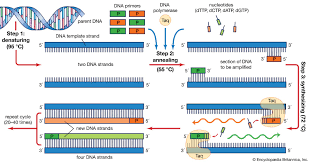
- Polymerase Chain Reaction (PCR) for rapid detection of resistance genes
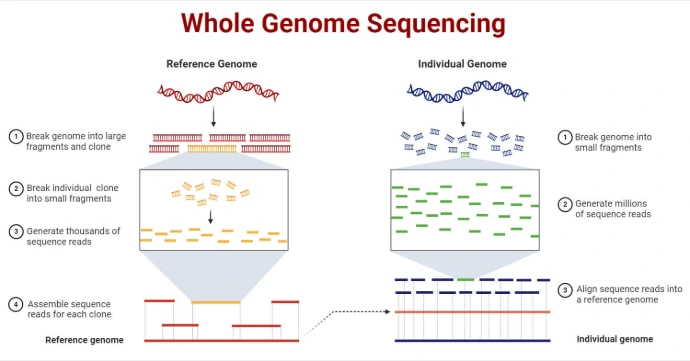
- Whole Genome Sequencing (WGS) for comprehensive analysis of bacterial genomes
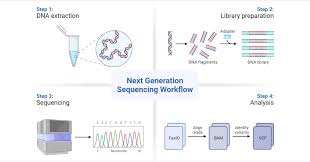
- Next-Generation Sequencing (NGS) for high-throughput identification of pathogens and virulence factors
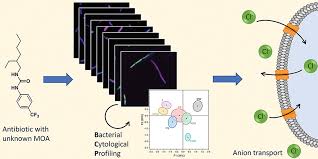
- Cytological profiling for rapid phenotypic antibiotic susceptibility testing
These tools enable clinicians to make informed decisions, reduce unnecessary antibiotic use, and improve patient outcomes.
01
Polymerase Chain Reaction (PCR) is a rapid laboratory technique that generates millions to billions of copies of a specific DNA segment, acting as a molecular photocopier
02
Whole Genome Sequencing (WGS) is a laboratory process that determines the entire DNA sequence of an organism's genome at a single time, covering both coding and non-coding regions. It provides a comprehensive, high-resolution view used to identify inherited disorders, track disease outbreaks, analyze cancer mutations, and advance personalized medicine.
03
Next-Generation Sequencing (NGS) is a high-throughput, massively parallel technology that sequences millions of DNA or RNA fragments simultaneously. It enables rapid, cost-effective, and detailed analysis of entire genomes, transcriptomes, or targeted regions, revolutionizing personalized medicine, cancer diagnostics, and genetic research.
Staphylococcus aureus and Antibiotic Resistance
Staphylococcus aureus is a major opportunistic pathogen responsible for a wide range of infections, from mild skin conditions to life-threatening systemic diseases. Its ability to produce diverse virulence factors and develop resistance to multiple antibiotics makes it particularly challenging to manage.
Methicillin-resistant Staphylococcus aureus (MRSA) is one of the most well-known resistant pathogens. It has evolved to adapt to various environments, including hospitals, communities, and livestock.
Key features of S. aureus include:
- Production of toxins and enzymes that facilitate tissue invasion
- Ability to form biofilms
- Rapid acquisition of resistance genes
- Regulation of virulence through complex genetic systems
New therapeutic strategies focus on targeting virulence factors rather than bacterial viability, offering a promising approach to reduce resistance development.

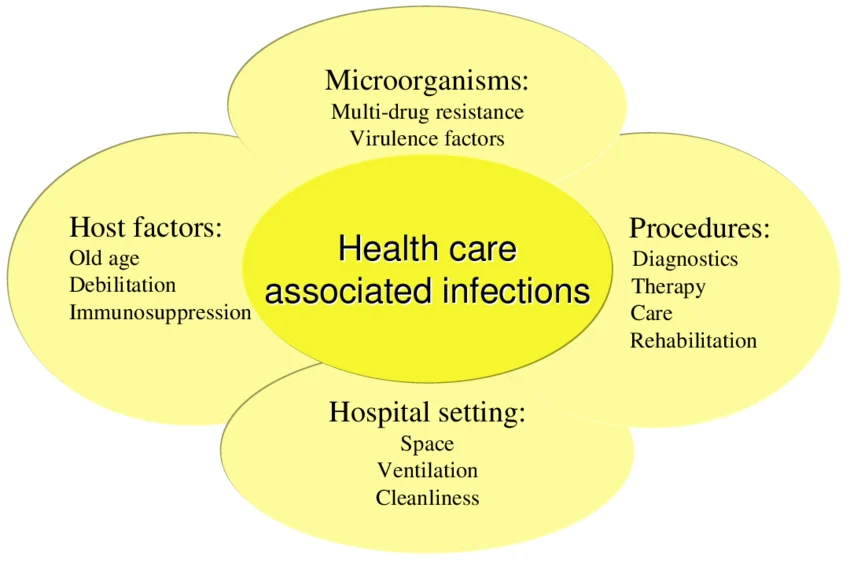
Healthcare-Associated Infections and Control Measures
Healthcare settings play a critical role in the spread of antibiotic-resistant bacteria. Poor infection control practices can lead to outbreaks of resistant pathogens, including:
- Methicillin-resistant Staphylococcus aureus (MRSA)
- Vancomycin-resistant enterococci (VRE)
- Clostridium difficile
- Carbapenem-resistant Enterobacteriaceae (CRE)
Effective infection control measures include:
- Strict hand hygiene protocols
- Patient screening and isolation
- Environmental cleaning and disinfection
- Antimicrobial stewardship programs
Studies have shown that implementing multimodal strategies significantly improves compliance with infection prevention guidelines and reduces transmission rates.
Conclusion
Antibiotic resistance is a multifactorial and evolving challenge that threatens the effectiveness of modern medicine. The increasing prevalence of multidrug-resistant bacteria, combined with the limited availability of new antibiotics, underscores the urgency of developing innovative solutions.
Key priorities include:
- Strengthening global surveillance systems
- Promoting responsible antibiotic use
- Advancing research in alternative therapies
- Enhancing infection prevention and control measures
- Leveraging new diagnostic technologies
A comprehensive and coordinated approach involving healthcare professionals, researchers, policymakers, and the public is essential to mitigate the impact of antibiotic resistance and ensure sustainable healthcare outcomes.